Jump to section:
UAccess Research Electronic Proposal Routing
Using UAccess Research
Supplemental Information
Training
Email Lists
UAccess Research Electronic Proposal Routing
UAccess Research is the University of Arizona's electronic proposal submission and award management system. It facilitates the preparation and processing of Pre- and Post-award tasks electronically, reducing the need for paper transactions. All proposals for external funding are submitted and routed for review/approval by the Principal Investigators/Co-Principal Investigators, Departments, Colleges, and the Sponsored Projects & Contracting Services office using UAccess Research. A UA NetID and password is required to log in to the system. You can log in by clicking on the Research link on the UAccess page.
Recommended Browsers:
- Google Chrome
- Mozilla Firefox
Using UAccess Research
User Guide: This guide provides field definitions and overall guidance about system navigation, proposal creation and routing in UAccess Research - Instructions for Proposal Initiators (updated February 18, 2022!). The UAccess Research Route Log Guide offers explanation specifically useful for understanding information in the route log for proposals.
Provide Access to a Specific Proposal ONLY: To provide view or edit access to a specific proposal development document, see the Proposal Access Roles Guide. Please note that while all roles can be granted prior to routing, once a proposal is submitted to routing only view access can be provided.
Request Privileges: You can use AccessFlow to request privileges to view proposals and awards at a Department or College level. This provisioning tool is also used to establish workflow for College and Department level approvals. Please see the UAccess Research Support, How-To's & Info and the UAccess Research Proposal Provisioning Guide.
UAccess Proposal Data Guide: The attached UAccess Proposal Data Guide is a two-page quick reference tool for initiators entering data in the UAR Proposal Development document to provide reminders and tips about the review, correction and finalization of proposal data. The guide includes hints about what changes require re-routing for updated approvals if changes are made while the proposal is already routing for approvals.
Reviewing and Approving Proposals: Prior to submission, proposals must be reviewed and approved using UAccess Research.
- Instructions for PI, Co-PI, Co-I and Key Person Review and Approval
- Instructions for Responsible Unit Review and Approval (Department, College, Other)
Request New Sponsor(s): If you cannot locate your sponsor in the sponsor table, please complete a New Sponsor Request form as soon as possible. The form was updated in January 2024 and is now web-based, and will notify Sponsored Projects Services upon completion. Remember to include as much identifying information as available. Whenever possible, the New Sponsor Request form should be submitted prior to routing the UAccess Research proposal document. To start a proposal document without the new Sponsor Code, please use the "Miscellaneous Sponsor" (Sponsor Code: 009800) as a placeholder. Once Sponsored Projects Services has created the new sponsor, you will get an email letting you know that it is available. If you have not already begun routing the proposal document, remember to replace the "Miscellaneous Sponsor" with the one just created.
Frequently Asked Questions for Early Investigators
Not finding what you're looking for? Try searching all research-related FAQs.
When submitting a proposal that may require additional space, renovations, or enhancements to utilities (like electrical, plumbing, or mechanical systems), it is crucial for investigators to reach out to Planning, Design & Construction (PD&C) and/or Facilities Management (FM) three months prior to the submission deadline. Such projects can involve significant costs and require additional lead time. By consulting the appropriate units well in advance, investigators can obtain cost estimates to include in their proposal budget whenever possible, and/or request and obtain commitments for alternate sources of funding prior to proposal submission. This ensures that the necessary funds and approvals are in place when awards are received so they can be accepted without delay.
Answer this question Yes if additional space, renovations, or changes in electrical, mechanical, or plumbing systems will be needed to conduct the project.
When answered Yes, a Questionnaire for "Space, Renovation" will be created requiring answers to additional questions:
What is the proposed location of additional space, renovations, or changes in electrical, mechanical, or plumbing systems? Be specific to avoid proposal delays (e.g. building name, room, total square feet, potentially impacted utilities systems, estimated cost, etc.).
In the text box, provide a brief summary of the anticipated needs. Be specific to avoid proposal delays and at a minimum provide the following: building name, street address, and room number if applicable; total square feet; potentially impacted utilities systems; cost estimate for changes.
Is the full cost of space changes or renovations included in the proposal budget?
Answer this question Yes if you have obtained a cost estimate and the full costs of the estimate are included in the proposal budget.
Answer this question No if you have not included the full costs of the cost estimate in the proposal budget or you have not obtained a cost estimate.
If not, What is the source of funds for space costs not included int he proposal?
If you answered No to the preceding question, in the text box enter specific amount(s), account number(s), and department(s)/unit(s). Documentation of alternate approved source(s) of funds should be attached in the Attachments section of the UAccess Research proposal.
Additional, more detailed guidance on estimating square footage, identifying which unit a space is assigned to, obtaining renovation estimates, submitting space change requests, and more can be found here.
Answer this question ‘Yes’ if this proposal involves companies or non-profits as partners if they are not already listed as a sponsor or prime sponsor. This would be the case for proposals where a company or nonprofit is a subrecipient, consultant, advisor, etc. If the answer is ‘Yes’ then it is required in the UAccess Research (UAR) proposal to list the full name(s) of any company or non-profit organization that will be involved as a partner, separated by commas.
Answer this question ‘No’ if the partner is already captured in the UAR proposal as the sponsor or prime sponsor.
Company and non-profit information helps RII to identify researchers, companies and non-profits interested in partnerships to strengthen responses to future sponsorship opportunities Contact Research Development Services (RDS) at ResDev@email.arizona.edu(link sends e-mail) for additional guidance about this question.
Answer this question ‘Yes' if the proposal involves handling any human derived products (tissue, saliva, organ, semen, vaginal secretions, feces, urine, blood, cells, etc.), or any other potential infectious materials (PIM) that could carry and transmit bloodborne pathogens. For assistance, contact Research Laboratory & Safety Services at (520) 626-6850 or rlss-bio-support@email.arizona.edu(link sends e-mail) or visit the or visit the Recombinant and Biohazardous Materials web page.
As a National Cancer Institute Designated Comprehensive Cancer Center Facility, any and all use of the facilities of the Cancer Center must be reported to the National Institutes for Health (NIH) on an annual basis. This includes any use of Cancer Center Facilities, including use of the Common Equipment Rooms and/or the Shared Services of the Cancer Center. use of any lab, office, common equipment or shared services or any part of the Cancer Center Facility requires approval.
Answer this question ‘Yes’ for any proposal that involves research related to cancer. This information is compiled for the Arizona Cancer Center annual report.
This question was added to the UAccess Research (UAR) proposal in April 2020 to easily track and report on sponsored project proposals and awards related to COVID-19. Is the project scope of work related to COVID-19?
If you have any questions please contact sponsor@arizona.edu(link sends e-mail).
Enter as many Facilities & Administrative rates as will be applicable to a proposal separated by a forward slash and without the "%" symbol. Example: 53/53.5
If you have any questions please contact sponsor@arizona.edu(link sends e-mail).
Select the appropriate rate category from the drop-down list in the UAccess Research (UAR) proposal. Visit the Facilities & Administrative (F&A) Rates page of the Research Support website and view the F&A rate table for additional guidance.
For further questions, please contact sponsor@arizona.edu(link sends e-mail).
Rate Categories:
- Federal negotiated rate: It is the University of Arizona’s policy to request the appropriate federally negotiated F&A rate for all sponsored activity regardless of funding source, the federally negotiated F&A rate will be the basis for budget purposes on all sponsored activity. When funding flows from a federal prime sponsor to the University of Arizona, through a pass-through entity, as in the case of federal flow-through awards, the University will accept stipulations that meet the requirements of Uniform Guidance 2 CFR 200.414
- Sponsor F&A rate stipulation: If a sponsoring agency limits or forbids the reimbursement of F&A based on federal/state/local law, administrative regulation, or published sponsor policy, exceptions for Federal Flow through or Stipulated F&A Rates apply. Documentation for a stipulated rate must be included with the proposal. To document a stipulated rate, attach a copy of the law, regulation or published policy stating the limitation on F&A reimbursement under the Attachments section of the UAccess Resarch proposal document as attachment type "F&A Stipulation".
- Other standard UA F&A rate: Other standard UA F&A rates are described in the standard F&A rate table on the Facilities & Administrative (F&A) Rates page.
- F&A waiver (rarely approved, attach waiver request template): The decision to grant or deny a waiver request is at the sole discretion of UArizona Research Innovation, & Impact (RII) but is in concurrence with the approval from the representative Department Head, and Dean or Associate Dean for Research. These offices will work with RII to review and obtain the approval in advance of a UAR application or contract. All requests must use the F&A Waiver Request Template with the Department Head and Dean or Associate Dean for Research approval and must be submitted with a UAR proposal no less than five working days in advance of the deadline. Attach the Template in the UAR proposal Attachments section as attachment type "F&A Waiver Request Template."
As of July 2024 this UAccess Research (UAR) proposal option is no longer in use.
If you have any questions please contact sponsor@arizona.edu(link sends e-mail).
FDA/EPA Quality Assurance (GLP/cGMP/QA)?
Answer this question ‘Yes’ if the proposal involves any of the terms below or requires adherence to any FDA/EPA quality assurance program. If the project you are proposing involves the manufacture of a medical/therapeutic product, the evaluation of an FDA grant “Test or Control Article”, or collaboration with another researcher/institution upon research requiring FDA/EPA quality assurance program enrollment please contact Research Laboratory & Safety Services at rlss-help@arizona.edu. Visit the Good Laboratory Practices web pages for additional guidance.
At this time, the RLSS maintains a voluntary GLP preparation program (training & inspections), the UArizona has not constituted an institutional Quality Assurance Unit and would need reasonable time to do so to accommodate anyone with an FDA/EPA quality assurance program requirement.
- Good Laboratory Practices
- Good Manufacturing Practices
- Quality Assurance
- 21CFR
- Title 21
- EPA Quality Program
- GLP
- GMP
- cGMP
- Test Article
- Control Article
Enter the account number of a past sponsored project if the current proposal may be funded as a new and separate award but is the next phase or a continuation of the past project.
If you have any questions please contact sponsor@arizona.edu(link sends e-mail).
Answer this question ‘Yes’ if any portion of the project is to be completed with the assistance of collaborators in a foreign nation and use the lookup feature in the UAccess Research (UAR) proposal to select the country of each foreign collaborator.
Answer this question ‘Yes’ if the proposal involves the use and storage (including cleaning and sterilizing of equipment) of any quantity of any solvents, oxidizers, corrosives, compressed gases, cryogenics, heavy metals, dust-generating compounds, ATF regulated materials, DEA Controlled Substances, pesticides, fertilizers, and/or hormones/steroids. Visit the Chemical Safety Program web page for additional guidance or contact Research Laboratory & Safety Services at rlss-help@arizona.edu(link sends e-mail) with questions.
Examples include but are not limited to:
- Ethanol
- Methanol
- Tetrahydrofuran
- Acetone
- Potassium permanganate
- Hydrogen peroxide
- Sodium/Calcium hydroxide
- Sulfuric/Nitric/Acetic acid
- Carbon dioxide
- Argon
- Liquid nitrogen
- Arsenic
- Mercury
- Mine tailings
- Silica powder
- Praxair FE-271 (or other brand additive manufacturing metal powder product)
- Nitrotriazolone (NTO)
- Nitrocellulose
- Ketamine
- Testosterone
- Cannabidiol (CBD)
- Round-Up (or other brand glyphosate product)
- Microthiol (or other brand sulfur fungicide product)
- Pyrethroids
- Novel mode-of-action insecticide
- Urea
- Mono-ammonium phosphate (11-52-0)
- Iron/Zinc/Molybdenum chelates
- Zoetis (or other brand estradiol product)
- Corticosteroids
Answer this Hispanic Serving Institution (HSI) question 'Yes' if the proposal falls into one of the three categories described below.
HSI Grant requests for proposal (RFPs) generally fall into three categories:
HSI Required: These funding opportunities are exclusive to HSIs and require proof that the university is an HSI. A digital copy of proof from the U.S. Department of Education can be provided upon request. Direct inquiries to Riley McIsaac, rmcisaac@arizona.edu.
Minority Serving Institution (MSI) Required: These funding opportunities are not exclusive to HSIs, but instead require that a submitting institution have MSI status (HSIs are included under the umbrella of MSIs). Proof of HSI status may or may not be required. Minority or underrepresented students (e.g., first generation, Pell grant recipient/low income) and/or communities/populations are often expressed as the targeted populations of interest.
Intentionally Involves Minority Students and/or Communities/Populations: These funding opportunities are not exclusive to HSIs or MSIs, but they explicitly call for or encourage the engagement of minority or underrepresented students, and/or communities/populations. Engagement of these students/communities/populations should be coupled with asset-based recruitment strategies, culturally relevant learning experiences, inclusive mentoring practices, and much more.
For questions or to learn more, please contact Riley McIsaac, Associate Director of Grants Development in the Office of Hispanic Serving Institutions (HSI) Initiatives (rmcisaac@arizona.edu).
Visit HSI Initiatives at: https://hsi.arizona.edu/.
The University of Arizona was federally designated as a Hispanic Serving Institution in Spring 2018, having reached the 25% undergraduate Hispanic enrollment requirement. UArizona was the first four-year public university in the state of Arizona to become an HSI and one of 16 R1 HSIs across the nation.
Answer this question ‘Yes’ if human subjects are involved in the proposal. The University is required to safeguard human participants that are involved in research projects. For any project involving the use of human participants, a protocol must be submitted to the University’s Human Subject Protection Program (HSPP) and the Institutional Review Board (IRB) for review and approval.
The HSPP and IRB ensure that Human Subjects rights and welfare are protected, the risk and potential benefits are weighed accurately, subject selection is fair and that the participants have an informed consent. Approval is required before any work with Human Subjects is initiated.
Visit the Human Subject Protection Program section of the Research Support website for additional guidance.
Answer this question ‘Yes’ if live vertebrate animals are involved in the proposal. A protocol must be submitted for approval to the Institutional Animal Care and Use Committee (IACUC) before an award is made, but not at the time of proposal.
IACUC oversees the university’s animal care and use programs. This unit ensures the humane and ethical treatment of the animals used in research, testing and education. IACUC reviews all requests to use vertebrate animals to ensure compliance with federal regulations.
Principal Investigators who plan to use animal subjects as a part of their research should contact a member of the IACUC early in the project design stage to determine appropriate species as models for research and appropriate procedures to be used in the course of research (from UHAP). Visit the Institutional Animal Care and Use Committee (IACUC) program pages of the Research Support website for additional guidance.
There are specific requirements for NSF proposals that may impact the resources or interests of a federally recognized Tribal Nation. Detailed guidance and FAQs for the University of Arizona process for NSF proposals can be found here.
Answer the Native or Indigenous research and/or engagement question ‘Yes’ if this proposal falls into one of the categories described below:
- the research or institutional engagement intentionally involves participation by members of a sovereign tribe or indigenous community and may foreseeably result in research results with implications specific to a tribe, indigenous community, or to individuals as members of the tribe or community. Note: Such engagement may occur with native or indigenous peoples outside the U.S.
- the research or institutional engagement takes place in Indian Country, or Alaska Native homelands, and/or on land under the control or jurisdiction of a sovereign tribe or indigenous community. Note: Such engagement may occur with native or indigenous peoples outside the U.S.
- human research is conducted in Indian Health Service (IHS) facilities or involving IHS staff or resources. Note: Additional engagement with the IHS, tribal Institutional Review Boards (IRBs), or other entities may be required.
- the research involves human subjects, including genetic testing or testing of blood, tissue, or other biological materials if the individual's membership in or affiliation with a tribe or indigenous community is identified, and that is intended to or may foreseeably result in conclusions or generalizations about a tribe, indigenous community, or individuals as members of the tribe or community.
- any research or institutional engagement involving human remains, funerary objects, sacred objects, or objects of cultural patrimony that are subject to the Native American Graves Protection and Repatriation Act.
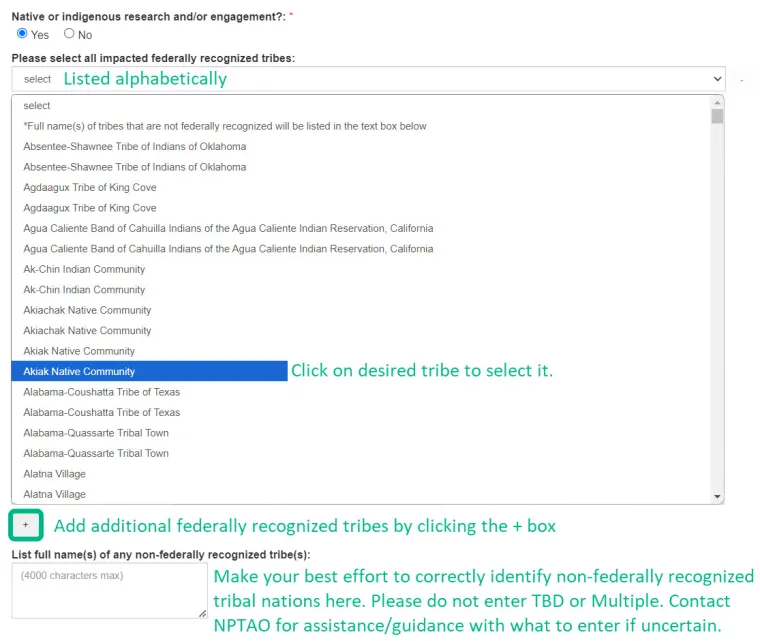
If the answer is 'Yes,' then you must also enter the full name of any tribe(s) that will be involved.
- Federally recognized Tribal Nations should be selected/added from the drop-down provided. You may add additional federally recognized Tribal Nations by clicking the + box below. You may remove added federally recognized Tribal Nations by clicking the - box to the right of the Tribal Nation.
- Other Tribal Nations and indigenous communities (including those external to the United States) should be entered in the text box provided. Please be aware that this information is used for reporting, so your best efforts at providing useful, accurate information are appreciated. You can reach out to Claudia Nelson for assistance/guidance with what to enter in this text field.
For questions or to learn more, please contact Claudia Nelson, Director, Native Peoples Technical Assistance Office (NPTAO) in Research, Innovation & Impact (RII).
For more information and resources, visit the Research & Engagement page on the Native American Advancement Initiatives & Research (NAAIR) website.
As a core service unit, Native Peoples Technical Assistance Office (NPTAO) serves as a primary research and resource liaison for Native affairs for the Office for Research, Innovation, and Impact (RII). This includes the Human Subjects Protection Program and Sponsored Projects review for compliance with ABOR 1-118 Tribal Consultation Policy and the UArizona Tribal Consultation Policy.
Enter the primary location where project activity will take place in the format Bldg-Rm-etc. Example: USB-510-A
The on- or off-campus designation of a project is determined by the location of the research and the wages incurred by the project. Separate On and Off Campus rates will not be used for a single project. The Off Campus Rate should be used for projects that are not conducted at university owned or leased facilities. Off Campus projects must include more than 50% of the wages incurred at a non-UArizona owned or leased facility. If lease costs are charged directly to the project budget, the project will be classified as Off-Campus.
Specific questions may be answered by Sponsored Projects Services (sponsor@arizona.edu(link sends e-mail)) in conjunction with Financial Services Rate Studies (ratestudies@fso.arizona.edu).
Radiation (Radioactive Material, Sealed Source, Radiation Generating Machine)?
Answer this question “Yes” if this proposal involves the use of Radiation.
Your work will need to be approved by the UA Radiation Safety Committee (RSC) before you can start work. You can access the documents needed to submit to the committee at https://research.arizona.edu/radiation-safety-forms, “Application for (Radioactive Material/Radiation Machine/Sealed Sources) Approval.”
Training is required before any person can handle Radioactive Material, Radioactive Sealed Sources or Radiation Generating Machines. Radiation Safety Training can be found on https://edgelearning.arizona.edu/. If you have any questions please reach out to rlss-rad-support@email.arizona.edu.
Non-Ionizing Radiation (Laser)?
Also answer this question “Yes” if this proposal involves the use of any laser or laser product.
Your work will need to be reviewed and approved by the UA Laser Safety Officer (LSO) and Laser Safety Committee (LSC). You can access the documents needed to submit to the committee at https://research.arizona.edu/compliance/RLSS/radiation-safety/laser-safety-program/laser-approval.
Training is required before any person can operate a laser. Laser training (Laser Radiation Protection Course (LRPC)) can be found on https://edgelearning.arizona.edu/. If you have any questions please reach out to rlss-rad-support@email.arizona.edu.
Answer this question ‘Yes’ if the proposal involves any of the terms below or involves any biohazardous and/or recombinant materials. For assistance, contact Research Laboratory & Safety Services at (520) 626-6850 or rlss-bio-support@email.arizona.edu(link sends e-mail) or visit the Recombinant and Biohazardous Materials web page.
If the project you are proposing involves the handling or storage of Risk Group 3 biohazardous and/or recombinant materials that would require the use of a BSL-3 laboratory, please contact Research Laboratory & Safety Services at rlss-help@arizona.edu. BSL-3 work must be approved in advance by the Institutional Biosafety Committee (IBC). You can access the documents needed to submit to the committee at Institutional Biosafety Committee | UArizona Research, Innovation & Impact. Access and training for the BSL-3 spaces is a lengthy process, so reaching out to RLSS early in the process is optimal.
- Adenovirus
- Adenoassociated Virus
- AAV
- Cloning
- Gene therapy
- Genomic library
- Microarray
- Plasmid
- Probes
- Recombinant
- Retrovirus
- Restriction fragment polymorphism (RFLP)
- Sequencing
- Southern / Northern hybridization
- Transformed cells
- Vector
- Bordetella pertussis
- Campylobacter
- Cell Culture
- Coccidiodes
- Chlamydia
- Clostridium
- Coxiella
- Cryptosporidium
- Cyclospora
- Giardia
- herpes
- HIV
- Influenza
- Monkeypox
- Mycobacterium
- Yesinia pestis (plague)
- Salmonella
- Toxin
- Vaccinia
- SARS-CoV-2
- Risk Group 2
- Risk Group 3
- Virus
- Bacteria
- Fungus
- Parasite
- Rickettsia
This data point will help campus units more accurately calculate proposal success or win rates. Units want to know what percentage of proposals that are reviewed and competitively scored against others for an open funding opportunity are ultimately funded.
Examples:
Answer 'Yes' if the proposed project...
- is an incoming subaward which is part of a proposal submission going in for review with the prime proposal at another institution
- will be reviewed and might or may not be awarded based on merit or a competition
Answer 'No' if the proposed project...
- if UArizona knows in advance that funding will be awarded, a funding decision has already been made and UArizona employs a certain person with unique expertise to accomplish project goals
- is a subaward from another institution that already received a prime award and UArizona is being asked to complete part of the work afterward
- will definitely be funded because UArizona has particular professional expertise
- is an administrative supplement that requires an application but funding is pre-approved
If you have any questions please contact sponsor@arizona.edu(link sends e-mail).
If proposal activity will take place at Banner University Medical Center (BUMC) facilities, answer 'Yes' to the appropriate location. These fields are not required so no response is the same as a 'No' answer.
- Banner - University Medical Center Tucson
- Banner - University Medical Center South
- Banner - University Medical Center Phoenix
If a project involves work on a U.S. military base abroad, additional international travel insurance requirements may be required. For expert guidance, contact the UA Export Control Program well in advance of travel.
Defense Base Act (DBA) Insurance is a federal requirement for international travel that is associated with the U.S. federal government. There are two primary triggers when DBA must be obtained:
- Travel abroad as part of a public work or service contract with the U.S. federal government where the UA is a contractor or subcontractor
- Travel to conduct work on a U.S. military installation abroad
DBA is generally NOT required for travel under federal research grants, cooperative agreements with federal agencies, or other authorized university travel unless one of the two triggering criteria listed above are applicable.
To arrange DBA insurance, ask your departmental Business Officer to complete a DBA Insurance Application Form and submit it to Risk Management Services a minimum of 30 days in advance of departure. Email confirmation of DBA coverage will be sent to the traveler and their department. Contact Risk Management Services at (520) 621-1790 or risk@email.arizona.edu for additional forms information.
Training
Sponsored Projects & Contracting Services provides a number of training opportunities, both online and in person. Please visit the UAccess Research Training page to find out about instructor led workshops or view training videos.
Email Lists
RAMTALK List: RAMTALK is a listserv that notifies subscribers of funding announcements, policies, and procedures.
To subscribe to RAMTalk, you may:
- From your University of Arizona email, send an email to ramtalk@list.arizona.edu from the email address you wish to subscribe with the subject: subscribe ramtalk Firstname Lastname (indicate your own first and last name) and leave the message body blank.
or
- Go to https://list.arizona.edu/sympa and click UA NetID Login in the upper right corner. Once logged in, enter ramtalk in the search box and click Search lists. Click on the hyperlinked list and then click Subscribe in the left navigation menu. Enter your e-mail address and click submit.
To unsubscribe from RAMTalk, you may:
- From your University of Arizona email, send an email to ramtalk@list.arizona.edu with the subject: unsubscribe ramtalk and leave the message body blank.
or
- Go to https://list.arizona.edu/sympa and click UA NetID Login in the upper right corner. Once logged in, select ramtalk from your lists. Select Unsubscribe in the left navigation menu. Click OK to verify that you wish to unsubscribe.
Full instructions on Sympa email commands can be found here.

