Disclosure of Outside Interests, Foreign Interests, Institutional Financial Interests & Outside Activity
University Employees and University affiliates and associates, including Designated Campus Colleagues who Conduct Research at or under the auspices of the University, are responsible for making disclosures to the Responsible Outside Interests Resources. More information about UArizona's disclosure requirements can be found in the Conflicts of Interest & Commitment Policy.
If you are not able to locate a disclosure certification in eDisclosure, please contact us at coi@arizona.edu.
Who must disclose?
All University Employees must disclose:
- Substantial Interest
All Full-Time (0.50 FTE or greater) University Employees must disclose:
- Outside Commitments
- Outside Employment
- Substantial Interest
University Administrators must disclose:
- Institutional Interests
- Outside Employment
- Outside Commitments
- Substantial Interests
Investigators* must disclose:
- Significant Financial Interests
- Significant Personal Interests
- Foreign Interests
- Outside Employment
- Outside Commitments
- Substantial Interests
*Investigators who are University Administrators must also disclose Institutional Financial Interests.
A Substantial Interest is any nonspeculative pecuniary or proprietary interest, either direct or indirect, other than a remote interest held by you or your Relative. Remote interest is defined in A.R.S. § 38-502(10).
The term Relative is set forth in A.R.S. 38-503 and means one's spouse or domestic partner, child grandchild, grandparent, sibling and their spouse or domestic partner, half-sibling and their spouse or domestic partner, and the parent, sibling or child of a spouse or domestic partner.
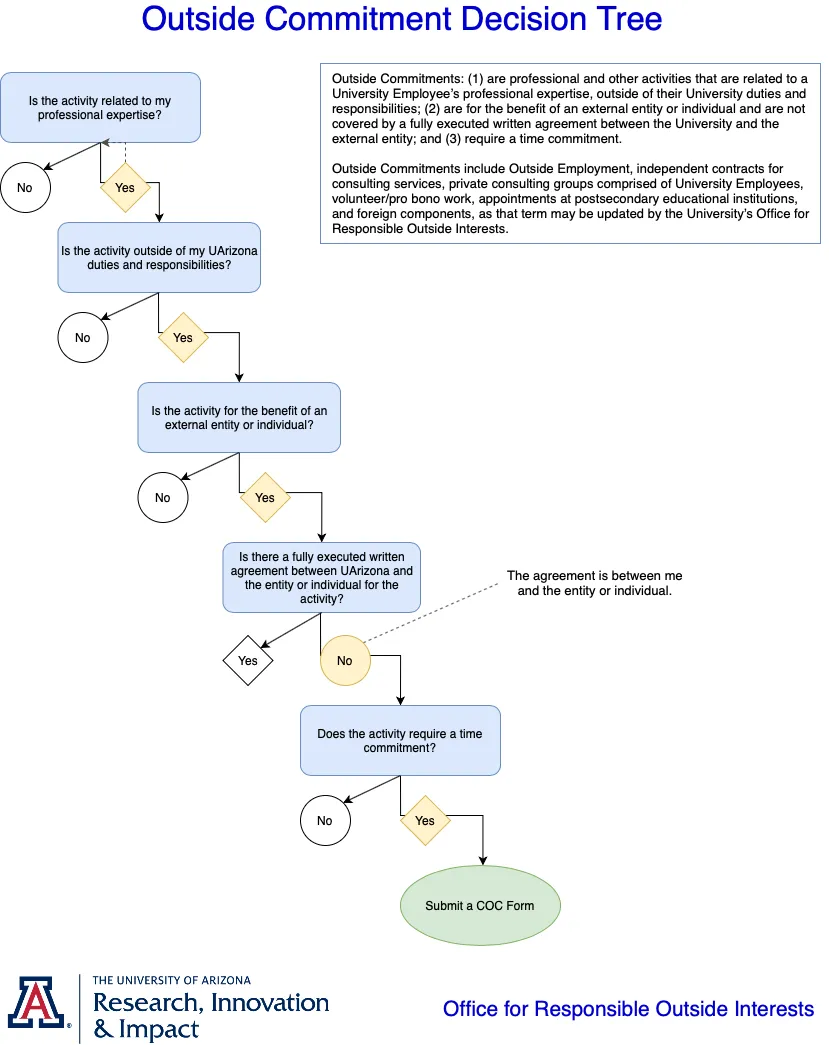
Outside Commitments (1) are professional and other activities that are related to a University Employee’s professional expertise, outside of their University duties and responsibilities; (2) are for the benefit of an external entity or individual and are not covered by a fully executed written agreement between the University and the external entity; and (3) require a time commitment.
Outside Commitments include Outside Employment, independent contracts for consulting services, private consulting groups comprised of University Employees, volunteer/pro bono work, appointments at postsecondary educational institutions, and foreign components, as that term may be updated by the University’s Office for Responsible Outside Interests.
University Administrator means any University Employee in a position of administrative leadership of a college, academic department, business, or other administrative unit, where a regular job requirement is to make institutional decisions on behalf of the University of Arizona.
The role of a University Administrator includes, but is not limited to, the following positions (whether such positions are staffed on an interim, full-time, or part-time basis):
- University Senior/Associate/Assistant Vice Presidents
- University Provost
- University Senior/Associate/Assistant Vice Provosts
- University Deans
- University Vice/Deputy/Associate/Assistant Deans
- University Directors
- University Department Heads/Chairs
- University Business Officers/Managers
- University Division Chiefs, Center Heads/Directors
- University Employees with the authority to sign agreements on behalf of the University of Arizona or Arizona Board of Regents
- University Employees whose duties and responsibilities include contracting or services related to Research administration, Research contracting, Research compliance, responsible conduct of Research, sponsored projects services, or technology transfer and who are in a position to influence decisions or commit University resources in the performance of the University Employee’s duties and responsibilities.
- Individuals who serve as Chairs on the University’s Institutional Review Board committees, regardless of whether such individuals are University Employees.
An Institutional Financial Interest is any of the following:
- A University Administrator’s Significant Financial Interest;
- A University Administrator’s Significant Personal Interest;
- A loan of any amount between a non-publicly traded entity and a University Administrator or their family member;
- The University’s equity or similar interest in a publicly traded entity valued at more than $100,000 or any amount in a non-publicly traded for-profit entity;
- Substantial gifts, including in-kind gifts, received from an actual or potential commercial research sponsor or an individual or entity that owns or controls products being studied or tested as part of the Research;
- Gifts totaling $1 million or more, cumulatively, and other gifts determined by the University’s Office for Responsible Outside Interests to be substantial;
- Any agreement with an external entity that includes the right for the University or a University Administrator or their family member to receive payments, royalties or other income from the commercial development or sale of an investigational product that is the subject of University Research;
- Ownership by the Arizona Board of Regents (ABOR), on behalf of the University, of a patent and/or other intellectual property right in or sponsorship of an investigational new drug (IND) or device (IDE) application with respect to any drug or device for which human subject Research will be conducted;
- Donation and sponsorship funds.
An Investigator is any person who shares the responsibility of Conducting Research. This includes, but is not limited to, the Principal Investigator (PI), Co-PI, Co-Investigator, Project Director (PD), Co-PD, Senior/Key Personnel, and any other person, regardless of title or position, who is responsible for Conducting Research performed by or under the auspices of the University.
More information on determining whether someone is an Investigator can be found here: Who is an Investigator?
A Significant Financial Interest is any one of the following interests, held by you or a Family Member (spouse, domestic partner, and/or dependent child), received in the past 12 months. All equity and compensation from an entity will be added together to determine if you meet the $5,000 threshold.
- Any income from Intellectual Property rights, such as patents or copyrights
- Any equity in a private company, regardless of value or percentage
- Equity valued at $5,000 or more in a public company (NASA Investigators only: Disclose equity in a public entity that represents more than 5% of the entity's equity, regardless of the value.)
- Compensation of $5,000 or more. This includes salary, consulting fees, honoraria, paid authorship, and other types of remuneration for services from an outside entity.
- PHS and Dept of Energy Investigators only: Disclose any reimbursed or sponsored travel related to your institutional responsibilities, regardless of the amount, unless received from a Federal, state, or local government agency of the United States; a domestic Institution of Higher Education; or a domestic research institute that is affiliated with a domestic Institution of Higher Education.
- NASA and NSF Investigators only: Disclose all Venture and Other Capital Financing.
- Salary, royalties, or other Remuneration paid by the University to a current University employee or appointee;
- Payments made by the University in exchange for the transfer of intellectual property rights assigned to the University and from agreements by the University to share any royalties related to such rights;
- Income from investment vehicles such as mutual funds and retirement accounts, as long as the Investigator does not directly control the investment decisions made by the investment managers within these funds or accounts; and
- Income from seminars, lectures, teaching engagements, or service on advisory committees or review panels sponsored by (i) a government agency (federal, state, or local); or (ii) an institution of higher education as defined at 20 USC § 1001(a); or (iii) an academic teaching hospital, medical center, or research institute that is affiliated with an institution of higher education.
Any managerial, professional, or Fiduciary Position you (or a Family Member (i.e., spouse, domestic partner and/or dependent child) hold in any outside entity, whether or not you or your family member is compensated. This can include officer, director, and board positions.
A Fiduciary Positions means one's legal and/or ethical obligation to act in the best interests (e.g., the financial and/or operating success) of another person or entity, regardless of whether such role is compensated. Examples of Fiduciary Positions include but are not limited to membership on a board of directors or board of advisors, or a management role in an entity (e.g., as a corporate officer, LLC member, general partner, and governing board member of a professional association).
Foreign Interests are:
- Participation in a foreign talent or similar-type program
- All resources and other support, both domestic and foreign, for ongoing research projects, including those conducted at a different institution
- In-kind contributions from domestic and foreign institutions or governments that support your research activities
- Any payment, reimbursement, travel support or other compensation, of any amount, that you personally receive, or will personally receive, from a foreign entity
What should I disclose in eDisclosure?
Please remember that COI Disclosures, except Foreign Interests, are also required for your relatives' interests. This table will be updated as federal regulations, state law and/or ABOR policies may be updated in the future.
| Activity Type | COI Disclosure | COC Disclosure Prior approval required if your UArizona FTE is 0.50 or greater | No Disclosure Required |
| Outside Employment with Remuneration less than $5,000 | x | ||
| Outside Employment with Remuneration of $5,000 or more | x | x | |
| Outside Commitment with no Remuneration | x | ||
| Outside Commitment with Remuneration less than $5,000 | x | ||
| Outside Commitment with Remuneration of $5,000 or more | x | x | |
| Editorial compensation of less than $5,000 | x Only if Outside Commitment | ||
| Editorial compensation of $5,000 or more | x | x Only if Outside Commitment | |
| Managerial, professional or Fiduciary Position (even if not compensated) | x | x Only if Outside Commitment | |
| Equity (shares, ownership, stock options) private company (any value) | x | ||
| Equity (shares, ownership, stock options) public company valued at less than $5,000 | x | ||
| Equity (shares, ownership, stock options) public company valued at $5,000 or more | x | ||
| Income from investment vehicles such as mutual funds and retirement accounts, as long as you do not directly control the investment decisions made by the investment managers within these funds or accounts | x | ||
| Consulting, advisory, or speakers bureau with no Remuneration | x Only if Outside Commitment | ||
| Consulting, advisory, or speakers bureau with Remuneration less than $5,000 | x Only if Outside Commitment | ||
| Consulting, advisory, or speakers bureau with Remuneration of $5,000 or more | x | x Only if Outside Commitment | |
| Intellectual Property rights (license / royalties of any amount paid directly to individual by an entity other than UArizona) | x | ||
| Remuneration (e.g., honoraria or other payments for services) or Reimbursements of less than $5,000 (excluding sponsored travel) | x Only if Outside Commitment | ||
| Remuneration (e.g., honoraria or other payments for services) or Reimbursements of $5,000 or more (excluding sponsored travel) | x | x Only if Outside Commitment | |
| Sponsored or reimbursed travel of any amount | x only if have PHS or Dept. of Energy Funding | ||
| Substantial Interest | x | ||
| Volunteer or pro-bono work that is unrelated to my professional expertise | x | ||
| In-kind contributions from domestic and foreign institutions or governments that support your research activities | x | ||
| All resources and other support, both domestic and foreign, for ongoing research projects, including those conducted at a different institution | x | ||
| Participation in a foreign talent or similar-type program | x | x | |
| Any payment, reimbursement, travel support or other compensation, of any amount, that you personally receive, or will personally receive, from a foreign entity | x | ||
Foreign Components (The performance of any significant scientific element or segment of a project outside of the United States, either by the recipient or by a researcher employed by a foreign organization, whether or not grant funds are expended.) Includes, but is not limited to, (1) the involvement of human subjects or animals, (2) extensive foreign travel by recipient project staff for the purpose of data collection, surveying, sampling, and similar activities, or (3) any activity of the recipient that may have an impact on U.S. foreign policy through involvement in the affairs or environment of a foreign country. | x |
A University Employee may obtain outside employment or render professional services provided (1) it does not in any way interfere with the person's university duties and the outside employment is fully consistent with all rules promulgated by UArizona and ABOR. Thus, UArizona implemented the now repealed Conflict of Commitment Policy in 2014. The standards of that policy are embodied in the Conflicts of Interest & Commitment Policy.
UArizona has a fiduciary duty to steward federal funds and ensure they are not used for the benefit of a third party.
A public employee shall not use or attempt to use their official position to secure any valuable thing or benefit for the employee that would not ordinarily accrue to the employee in the performance of the employee's official duties if the thing or benefit is of such character as to manifest a substantial and improper influence on the employee with respect to the employee's duties. A violation is a criminal offense.
Non-Financial Conflicts of Interest
Outside Commitments must also be reviewed to protect UArizona's Research enterprise. "Researchers who get federal funding have to watch out for 2 kinds of conflict of interest: financial and non-financial."
In a non-financial conflict of interest, an individual faces conflicting obligations from multiple employers or other entities. For example, an individual may be required to improperly share information with, or withhold information from, an employer or funding agency, which threatens research security and integrity.""
42 CFR 50, Subpart F, Promoting Objectivity in Research (FCOI Regulation)
Public Health Services FCOI Regulation
200 CFR 200, Chapter IX(A) Conflict of Interest Policies
National Science Foundation Grantee Standards
Department of Energy Interim Conflict of Interest Policy
Factors for Assessing Senior/Key Personnel Disclosures
Defense Advanced Research Projects Agency's Risk-Based Measures to Assess Potential Undue Foreign Influence, Conflicts of Interest or Conflicts of Commitment
Federal Acquisition Regulations - Organizational and Consultant Conflicts of Interest
Guidance for Implementing NSPM-33
"Recently, the federal government determined that disclosure requirements should be standardized across all funding agencies. It also indicated that federal funding agencies ""should require that recipient organizations instruct covered individuals on how to disclose information related to potential financial conflicts of interest, including but not limited to: private equity, venture, or other capital financing.
To ensure UArizona employees are in compliance with federal regulations and requirements, UArizona applied a single standard for disclosure to all Investigators in 2014 when it implemented the now repealed Individual Conflict of Interest in Research Policy. That same philosophy and the same thresholds are embodied in the Conflicts of Interest & Commitment Policy."
An employee of a public agency who has, or whose relative has, a substantial interest in any contract, sale, purchase or service to or decision of such public agency shall make known that interest in the official records of such public agency and shall refrain from voting upon or otherwise participating in any manner as an officer or employee in such contract, sale, purchase or decision. A violation is a criminal offense.
"Federally-funded researchers may face conflicts of interest if they receive funding or other benefits from foreign entities, which may be looking to gain access to that research."
A regent or employee may not use or attempt to use his or her official position to secure any valuable thing or benefit that would not ordinarily accrue in the performance of his or her official duties, if the thing or benefit is of such character as to manifest a substantial and improper influence on the regent or employee.
A public officer or employee shall not use or attempt to use the officer's or employee's official position to secure any valuable thing or valuable benefit for the officer or employee that would not ordinarily accrue to the officer or employee in the performance of the officer's or employee's official duties if the thing or benefit is of such character as to manifest a substantial and improper influence on the officer or employee with respect to the officer's or employee's duties.
Reviewing & Accepting Gifts" Acceptance of some gifts are subject to Research Compliance review, including but not limited to: Conflict of Interest, Export Compliance and Human Subject.
The university must identify, on a case-by-case basis, individual and institutional conflicts of interest and conflicts of commitment that may arise as a result of the proposed transfer of intellectual property.
| Activity Type | COI Disclosure | COC Disclosure Prior approval required if your UArizona FTE is 0.50 or greater | No Disclosure Required |
| A loan of any amount between a non-publicly traded entity (i.e., a private loan) and a University Administrator or their family member | x | ||
| A loan of any amount between a publicly traded entity (i.e., a commercial loan) and a University Administrator or their family member | x | ||
| UArizona's equity or similar interest in a publicly traded entity valued at more than $100,000 or any amount in a non-publicly traded for-profit entity | x | ||
| Gifts totaling $1 million or more, cumulatively | x | ||
| Substantial gifts, including in-kind gifts, received from an actual or potential commercial research sponsor or an individual or entity that owns or controls products being studied or tested as part of the Research | x | ||
| Donation and sponsorship funds | x | ||
| Any agreement with an external entity that includes the right for the University or a University Administrator or their family member to receive payments, royalties or other income from the commercial development or sale of an investigational product that is the subject of University Research | x | ||
| Ownership by the Arizona Board of Regents (ABOR), on behalf of the University, of a patent and/or other intellectual property right in or sponsorship of an investigational new drug (IND) or device (IDE) application with respect to any drug or device for which human subject Research will be conducted | x |
|
Consult this decision tree to determine if you must submit a COC form